Cornell Study Uncovering DNA Impact of Chemotherapy Agent
New Cornell research sheds new light on how etoposide, a common chemotherapy agent, stalls and poisons the essential enzymes that allow cancer cells to thrive.
The findings from Michelle Wang’s lab, the James Gilbert White Distinguished Professor of Physical Sciences and Howard Hughes Medical Institute Investigator in the College of Arts and Sciences, will advance research into a variety of cancer inhibitors.
The group’s techniques will also enable the development of sensitive screening tools for identifying drug mechanisms that can improve patient treatment.
The study, “Etoposide Promotes DNA Loop Trapping and Barrier Formation by Topoisomerase II,” was published in Nature Chemical Biology on January 30.
Tung Le, a research specialist, and Meiling Wu, a postdoctoral researcher, are the co-lead authors.
ALSO READ: Pastor Describes Torture in Hell with Rihanna’s Music
For more than 40 years, etoposide has been used to treat a variety of cancers.
Etoposide works by targeting Type IIA eukaryotic topoisomerases, enzymes (also known as topo IIs) that allow cancer cells to replicate.
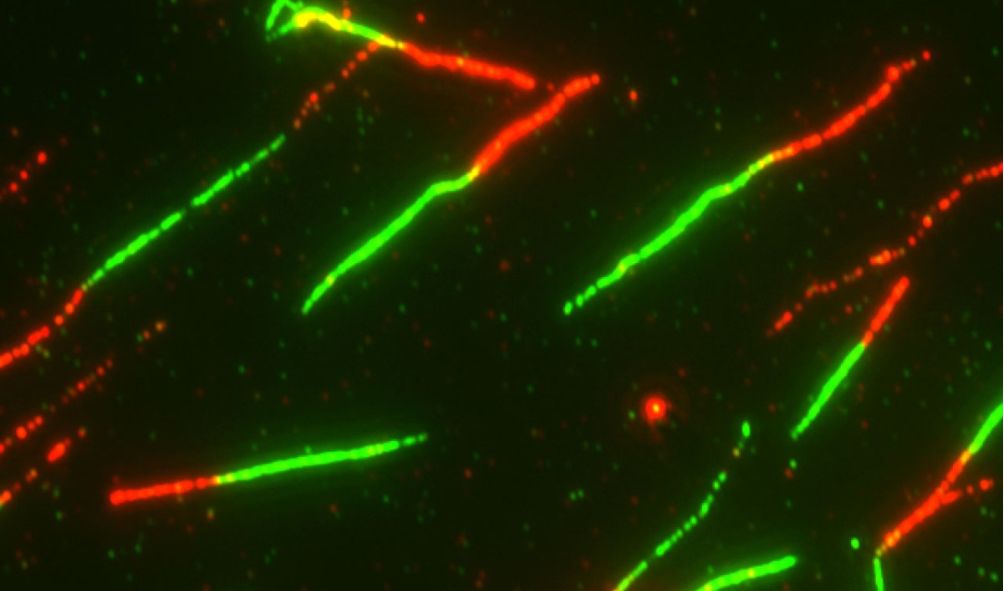
The long, entangled, helical-coiled strands of DNA are at the heart of the replication process.
Motor proteins must untangle, rotate, and copy these strands for cancer to spread.
Topo IIs are ideal for the job. They use an elaborate rope trick to relax the supercoiled DNA by cutting it, quickly passing another DNA strand through its middle, and reconnecting the cut DNA.
All of this is accomplished without causing damage to the DNA’s delicate genetic structure – an incredible, and incredibly fast, the feat of biology that occurs in the body approximately 300 billion times per day.
Etoposide’s main advantage is that it can stabilize a DNA double-stranded break before it is reconnected, preventing the cancer cell from replicating.
ALSO READ: Unlocking the Hidden Mysteries: The Surprising Power of Saliva
However, the complexities of how etoposide interacts with the structure of DNA have remained unknown.
Wang’s lab used three different single-molecule manipulation techniques to study the effect of etoposide on three topos IIs provided by collaborators led by Johns Hopkins University professor James Berger: yeast topoisomerase II, human topoisomerase II alpha, and human topoisomerase II beta.
“DNA topology is extremely difficult for people to grasp, both conceptually and in terms of torsional mechanical properties,” Wang said. “There were few ways to investigate it. But we just happen to have the right tools. And we have the right tools because we’ve been working on developing them for the last 20 years. These tools and this problem just happened to be in the right place at the right time.”
First, the researchers stretched DNA into various configurations with optical tweezers, demonstrating how etoposide compacts, releases, and breaks it, as well as create DNA loops.
This loop-trapping behavior astounded everyone because it revealed a previously unknown effect of etoposide.
It suggests that etoposide may stimulate topo II to significantly alter DNA structure and topology in vivo.
The researchers then used optical tweezers to unzip double-stranded DNA into two single strands for high-resolution mapping of protein interactions with the DNA, simulating the removal of a bound protein.
ALSO READ: Unlimited Children Policy Adopted in Sichuan, China
The findings suggest that etoposide can turn topo II into a powerful inhibitor of DNA-processing machinery.
Their third technique is a variation on magnetic tweezers in which they twisted DNA with a bound topo II and observed the topo II slowly relax the DNA.
They discovered that adding etoposide staggered this pattern, introducing pauses that correspond with the trapping of supercoiled loops.
The researchers now have a quantitative system for characterizing how other topoisomerase drugs behave after capturing the various ways etoposide enhances these actions and interferes with topo II function.
“I believe this provides us with a set of tools that will enable us to study many different types of topoisomerases and other types of drugs in a very comprehensive manner,” Wang said. “Everything we do is a simulation of what happens in the real world. We simply do it in a mechanically controlled manner. This is why it is so effective.”
Doctoral student Neti Bhatt and research specialist James Inman are among the co-authors, as are Johns Hopkins University School of Medicine’s James Berger and Joyce Lee.
The National Institutes of Health and the Howard Hughes Medical Institute funded the study.